What is cholangiocarcinoma?
CCA is a rare and aggressive type of cancer that forms in bile ducts. It is also called bile duct cancer. Bile ducts are thin tubes that carry a digestive fluid called bile from your liver to other digestive organs.
Most people with CCA do not have symptoms until the disease becomes more advanced. For this reason, it is often difficult for healthcare providers (HCPs) to diagnose the disease early.
While surgery may be used to remove the tumor in some cases, this may not be an option if the tumor takes up too much of the liver, or if the disease has spread to other parts of the body.
There are different kinds of CCA, based on where it is located.
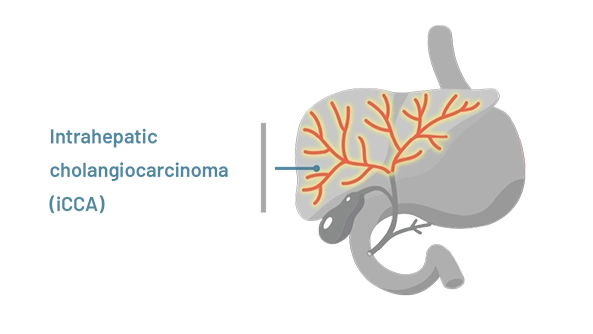
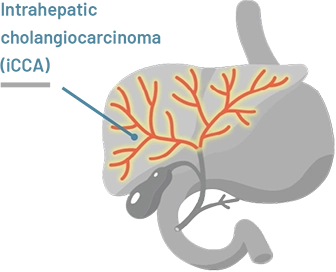
Intrahepatic (meaning inside the liver) cholangiocarcinoma starts in bile ducts within the liver
The location of cholangiocarcinoma may affect how HCPs treat the disease.
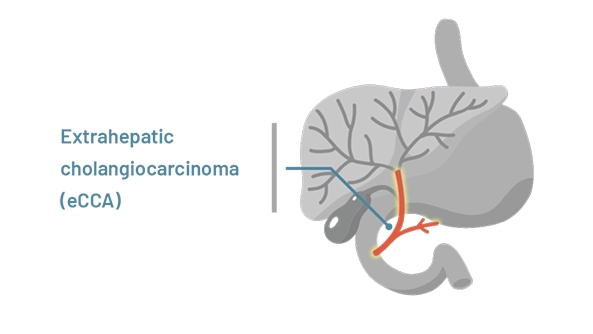
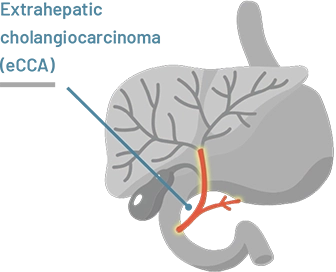
Extrahepatic (meaning outside the liver) cholangiocarcinoma starts in the bile ducts closer to the intestine
The location of cholangiocarcinoma may affect how HCPs treat the disease.
Causes of CCA
Cholangiocarcinoma occurs when the cells lining the bile ducts go through abnormal gene changes, which make the cells grow and divide without stopping. These cancer cells form tumors.
Over time, some cancer cells can break off and spread to other parts of the body. This is called metastasis.
There are different types of gene changes or defects that can lead to CCA, such as gene mutations, gene amplifications, or gene fusions.
Gene fusions occur when parts of two different genes abnormally join together
FGFR2 fusions
One specific type of gene fusion that can cause CCA is called a fibroblast growth factor receptor 2 (FGFR2) fusion.
- FGFR2 is a gene that makes the FGFR2 protein, which is involved in cell growth
- An FGFR2 fusion changes the FGFR2 gene and causes the cells in your bile ducts to make an abnormal version of the FGFR2 protein
- This abnormal FGFR2 protein can become overactive, causing these cells to grow and divide without stopping, which can lead to tumor growth in CCA
An FGFR2 fusion is one of the most common types of abnormal gene changes in people with iCCA.
NGS testing for FGFR2 fusions
After you are diagnosed with CCA, your doctor may order a test called next-generation sequencing (NGS), which is a type of molecular profiling, or biomarker testing.
This type of test can help determine if you have an FGFR2 fusion and could help direct how your disease is managed.
- In this test, HCPs examine a sample of the tumor to identify certain biomarkers, such as gene changes or defects that could drive tumor growth
- People may be eligible for PEMAZYRE if NGS testing identifies an FGFR2 fusion
It’s a good idea to talk to your healthcare provider about biomarker testing as soon as possible after your diagnosis.
Understanding molecular profiling in cholangiocarcinoma
Watch Dr Milind Javle, a gastrointestinal oncologist, explain molecular profiling—also called biomarker testing—and how it can help guide the way cholangiocarcinoma, or bile duct cancer, is managed.
This video is intended for informational use only, and is not designed to replace the medical advice of your healthcare provider (HCP).

What is the difference between “genetic” testing and “genomic” testing?
“Genetics” and “genomics” are two words that sound similar, but mean different things.
Genetics refers to the study of single genes and how they are involved in passing down traits from generation to generation. Genetic testing helps identify genetic conditions that may be inherited, like sickle cell anemia and cystic fibrosis.
Genomics is the study of how all of a person’s genes interact with each other and the environment. An FGFR2 fusion is an example of a genomic change that can cause cholangiocarcinoma.
Have you been prescribed PEMAZYRE?
Learn more about your dosing schedule, download resources to help you keep track of your treatment, and more.
Keep learning
The National Comprehensive Cancer Network® (NCCN®) is a not-for-profit alliance of 33 leading cancer centers devoted to patient care, research, and education. You can find in-depth explanations of CCA diagnosis and treatment in the NCCN Guidelines for Patients®.*
*Referenced with permission from the NCCN Guidelines for Patients® for Gallbladder and Bile Duct Cancers, 2023. © National Comprehensive Cancer Network, Inc. 2023. All rights reserved. Accessed July 22, 2024. To view the most recent and complete version of the NCCN Guidelines for Patients, go online to NCCN.org/patientguidelines. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
Have the conversation about PEMAZYRE today
Find out about PEMAZYRE—a therapy doctors have turned to for over 5 years to treat more than 1,500 patients with cholangiocarcinoma.†
†Commercially available in the US since 2020.


